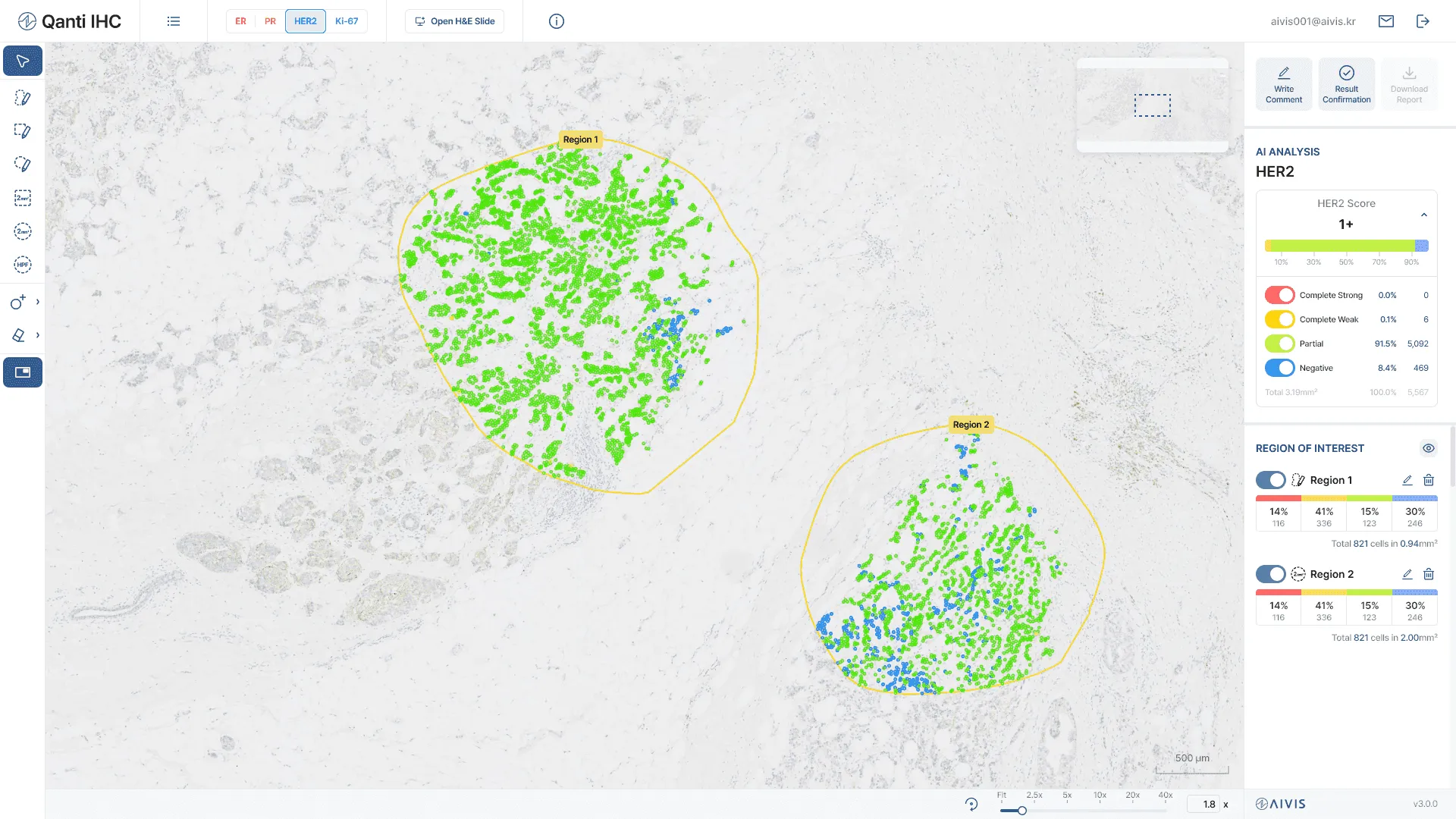
Description
- Qanti® Breast HER2 is designed to assist anatomic pathologists in the quantitative cell counts and semi-quantitative HER2 IHC score interpretation in breast cancer.
Scoring System
- Following ASCO/CAP 2023 and CAP 2025 Breast HER2 Interpretation Guidelines
- HER2 0 (null), 0+ (ultralow), 1+, 2+, and 3+
Performance
- Inter-observer agreement improvement: 0.4492 to 0.7524 (Fleiss' kappa), 0.7415 to 0.8849 (Kripendorff's alpha)
- Accuracy: 93.07%
Certified by MFDS(K-FDA)
Qanti® Breast ER, PR, HER2, and Ki-67 modules have been approved as 'Qanti® IHC' by the Korean Ministry of Food and Drug Safety.
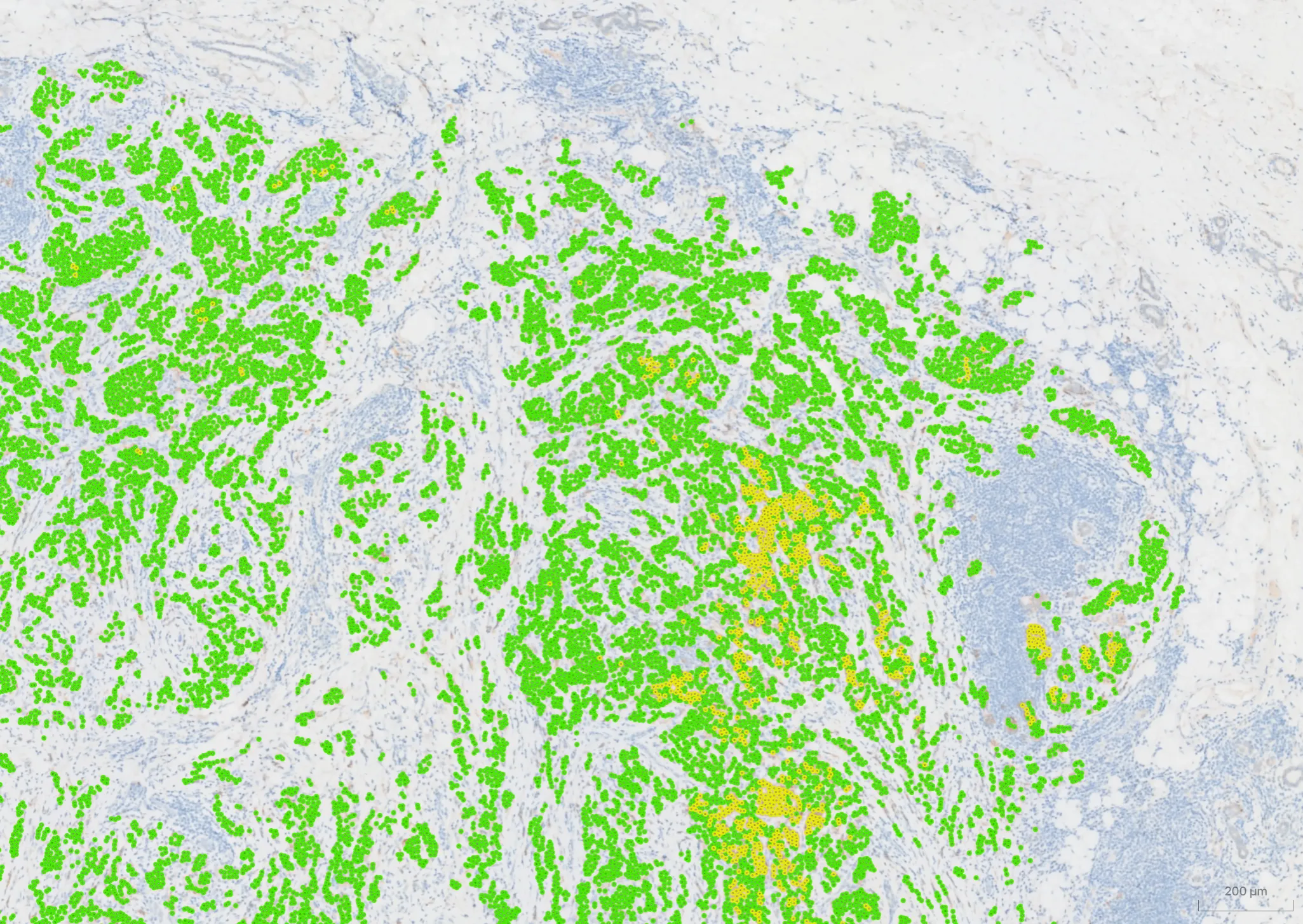
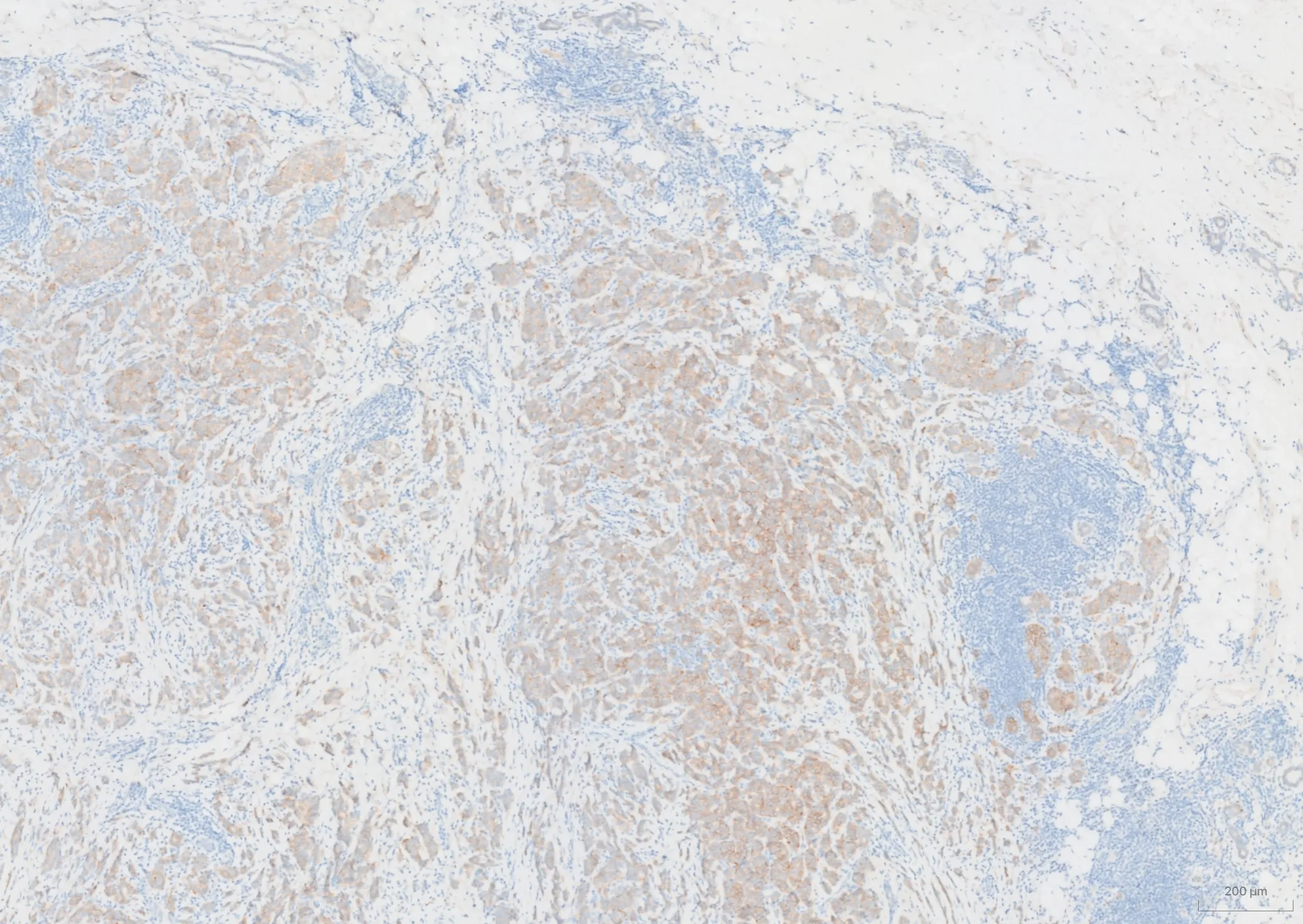
ESMO Asia 2025